|
6/17/2023 0 Comments Atomic emission spectrum 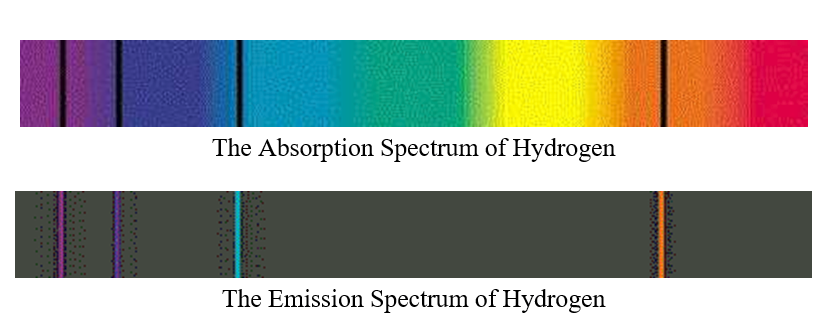
And finally potassium with the purple, or lilac shade.Īlright so if you took a prism to those actual the frame tests from those elements being burned or those compounds being burned. Then we have copper with its green color. 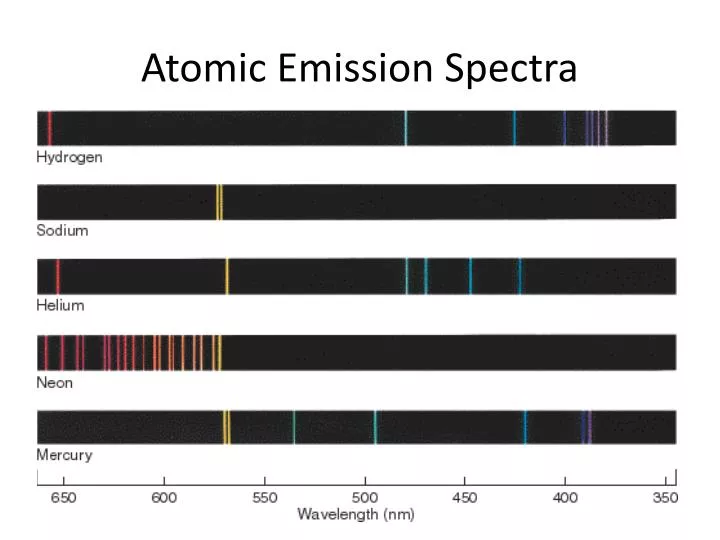
Next, we have sodium giving us a characteristic yellow flame. 
Not showing up very well here in the next one, but the red tints inside of that orange, there's some impurities there that is lithium. Then we have boron being green, strontium giving us the crimson red color. Then we have some barium chloride, with barium ions burning a dirty yellow color. So let's actually look at the video that shows this kind of information.Ī very dramatic version of the flame test demonstration here. So what happens when you have light emitted from different elements than when you burn them or when you actually light something that's actually not white light when you break kit up with a prism what that actually looks like. 
Alright so we know that when you take white light put it to a prism it breaks up into the rainbow the full visible spectrum. In the middle we have visible light and we're going to focus on visible light just for next couple of seconds. Alright so here is the overall general view of the electromagnetic spectrum going from high energy wave the gamma rays to very large energy waves the radio wave. Okay so first question is electromagnetic spectrum let's take a look at that. It's a set of frequencies of the electromagnetic spectrum emitted by excited elements of an atom. Positions are measured using the meter sticks, then wavelengths are determined from the positions using the graph itself or the equation of the best fit line for that graph.įor atoms that contain only one electron, the theory of atomic structure proposed by Niels Bohr can be used to calculate wavelengths for transitions between particular electronic energy levels of the atom.Alright so we're going to talk about atomic emission spectra. The calibration graph is therefore an integral part of the spectroscope. For example, using the same apparatus and without moving the relative positions of the meter sticks, diffraction grating and lamp, it is possible to view the spectrum of a new element, measure where its spectral lines occur on the meter stick, and then read the graph or use the equation of the line to determine the wavelength to which each of those positions corresponds. Once the best fit straight line has been determined, the equation of this line can then be used to convert positions of other spectral lines to wavelength. position of the spectral line will yield a straight line. Since this position depends upon the wavelength in a linear way, a graph of wavelength vs. Using a light source that contains known wavelengths of light, we can measure exactly where each known wavelength appears along a meter stick. As the light emerges after being reflected by the grating, these tiny lines cause the reflected light to interfere with itself in such a way that the different wavelengths of the light to appear in different positions to the left and right of the original direction in which the light was traveling. A diffraction grating is a piece of glass or clear plastic with many very narrow and closely spaced lines on it. If we view the light through a prism or a diffraction grating, however, the individual wavelengths are separated. To the naked eye, the various wavelengths (colors) of light emitted by an element are mixed together and appear as a single color that is a combination of the component colors. To measure these wavelengths in the laboratory, we must first separate them. Thus, the spectrum of an element can be stated by listing the particular wavelengths of light that its atoms emit. These two relationships combine to give a third: 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
